Industries that use these materials are concerned with relative humidity control in production and storage of such materials. Such substances include cotton, paper, cellulose, other wood products, sugar, calcium oxide (burned lime) and many chemicals and fertilizers. Many substances are hygroscopic, meaning they attract water, usually in proportion to the relative humidity or above a critical relative humidity. In human terms, our thermal comfort is in large part a consequence of not just the temperature of the surrounding air, but (because we cool ourselves via perspiration) the extent to which that air is saturated with water vapor. ( March 2021) ( Learn how and when to remove this template message)Īlthough the principles of psychrometry apply to any physical system consisting of gas-vapor mixtures, the most common system of interest is the mixture of water vapor and air, because of its application in heating, ventilation, and air-conditioning and meteorology. Unsourced material may be challenged and removed. Please help improve this section by adding citations to reliable sources. 
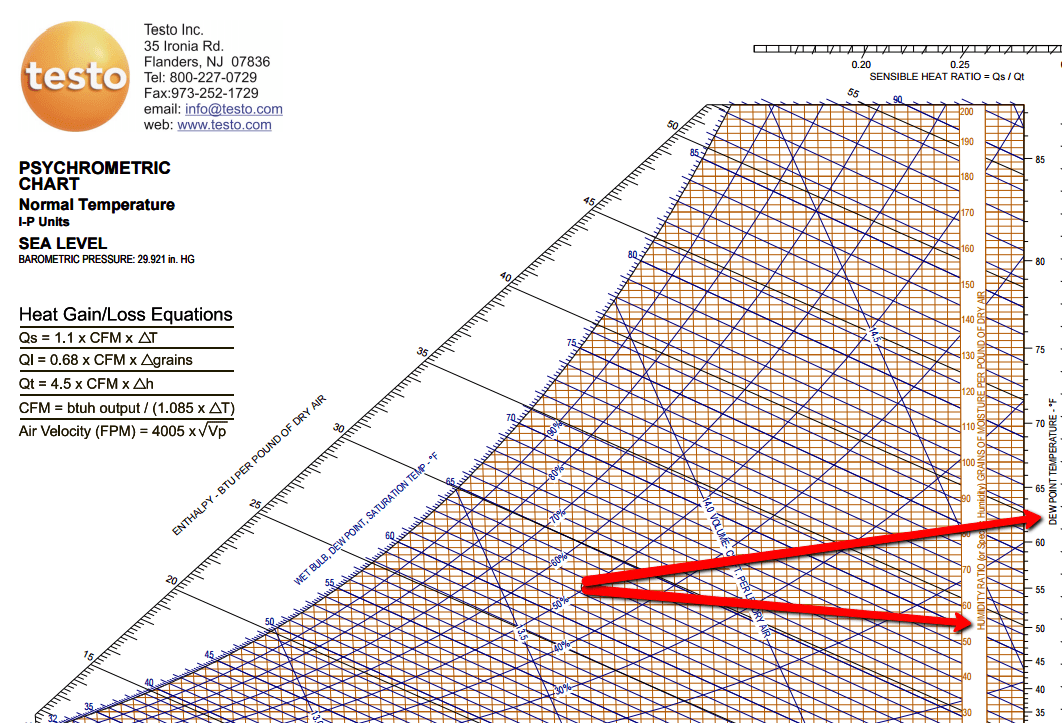
The above psychrometric properties can be calculated with the help of thermodynamic equations as stated below.Īssume the known properties are DBT, WBT & altitude(Sea-level) Standard pressue & temperature at the given altitude Values taken in this example are at sea-level)Īssume the below inlet and outlet air conditionsįor the above assumed values plotted psychrometric properties are Property (Verify that you have a right chart in place with respect to the altitude. With any two known values we can plot all other values using this chart with minimal approximation.īelow image is the representation of all the above thermodynamic properties plotted in a psychrometric chart. Psychrometric chart is a graphical representation of thermodynamic properties of air. > It is the pressure at which the air can have maximum water vapour for a given temperature. VP – Vapour pressure / partial pressure of water vapour (psi) 
> Pounds of water vapour per pound of air 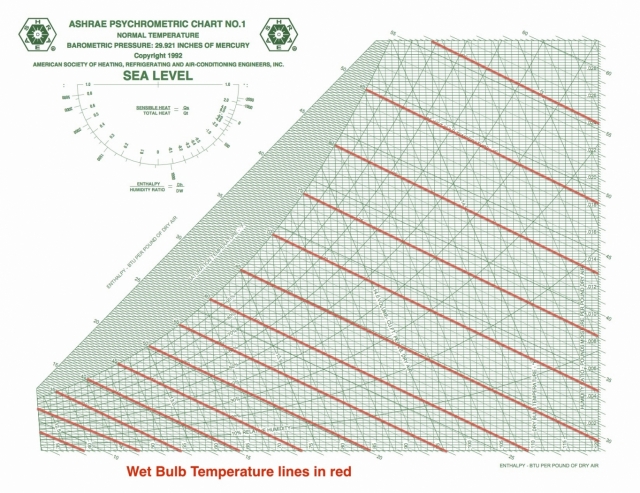
(OR) Specific humidity of air per specific humidity of saturated air.ĪH - Absolute humidity / humidity ratios / Specific humidity(lb/lb / gr/lb) (OR) Partial pressure of water vapour per equilibrium vapour pressure of water vapour at > Amount of water vapour per amount of saturated water vapour at a specified temperature Temperature at which humidity is 100% and air can not hold further water vapour. > Temperature at which the water vapour in the air condense in to water. > It is the thermodynamic temperature of air measured using a thermometer which is covered > It is the thermodynamic temperature of air measured using a thermometer freely exposed toĪtmosphere and shielded from radiation and moisture Resolve with Psychrometric chartīelow are the list of air properties we generally use in air conditioningįor simplicity consider there is no fresh air addition, AHU fan motor heat gain, and infiltration.Īlso a typical cooling and dehumidification process is considered in this example. Ĭlick here for Psychrometric calculator, computes psychrometric properties without chart and equations.Ĭlick here for Moisture content in air/Pychrometry, computes psychrometric properties without chart and equations. Psychrometric chart and related equations by Dasa. Study of Moisture content in air - Pychrometry Psychrometric chart and related equations Momentum equation in a 3D particle and its surface & body forcesĪir Pollution data-Telangana, Sanathnagar, HyderabadĪir Pollution data - Tamil Nadu Station: Alandur Bus Depot, Chennai - CPCB Law of Conservation of Momentum and Energyĭesign For Manufacturing and Assembly(DFMA) principles of Sheet Metal Work Law of Conservation of Mass and Continuity Equation Apparatus dew point temperature (ADP) & By-pass factor (BF)Īir Handling Unit (AHU) performance calculationĬhilled water flows measurement using Orifice plate, Balancing valves, Manometers & Pitot tubes. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
